At WiseGEEK, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What Is Tertiary Butyl Alcohol?
Tertiary butyl alcohol is a flammable and potentially toxic substance used in industrial processes. Molecularly it contains a methyl group, carbon, oxygen, and hydrogen. The compound is often used as an industrial solvent, as well as to boost octane levels in gasoline. It is also used for manufacturing perfumes, shellac, artificial leather, and photographic films. Safety measures must be adhered to when using tertiary butyl alcohol; it can be toxic if someone inhales or swallows the substance or gets it on his or her skin.
Workers who manufacture pharmaceuticals can be at risk for exposure to the substance. Production of safety glass, rubber and plastic cement, and paint removers are often other tertiary butyl alcohol industrial uses. It is typically flammable, either as a result of a spark, high temperatures, or direct exposure to flames. The boiling point is generally 180°F (82.4°C) and explosions can occur from just a trace amount of vapor.

Fires and explosions related to tertiary butyl alcohol can occur when it comes into contact with hydrochloric acid or oxidizing compounds. It can also decompose into toxic gases, so the compound should usually be stored in closed containers for safety reasons. Areas where it is used often need to be ventilated and protective clothing and equipment should generally be made available. A tertiary butyl alcohol material safety data sheet (MSDS) also suggests that the compound not be used with copper, alkali metals, or aluminum.

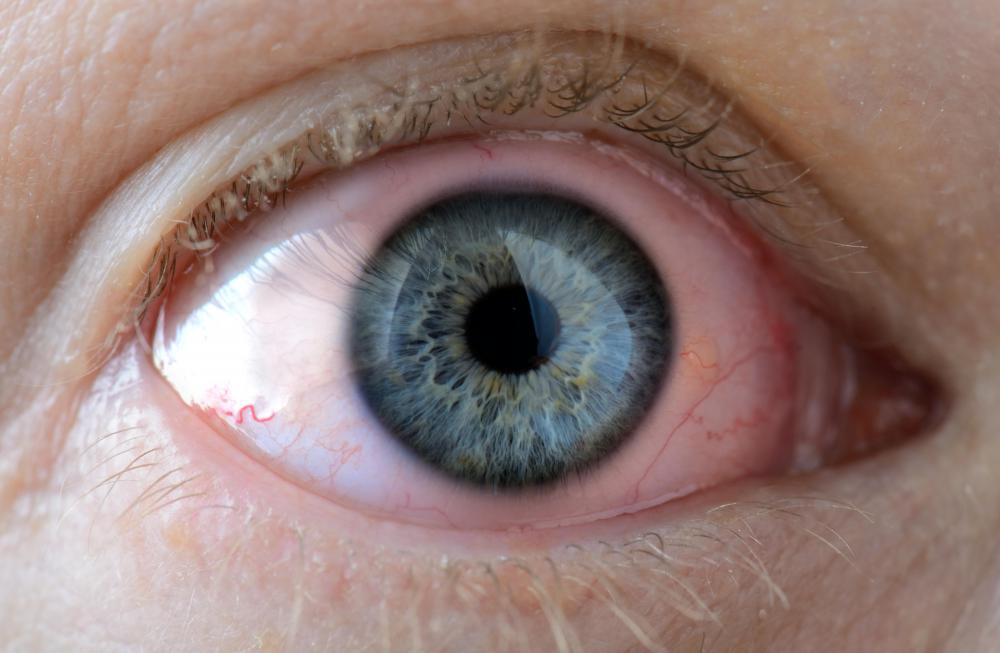
Exposure to tertiary butyl alcohol often causes irritation of the eyes, and irritation of the skin is also possible. Washing the affected area is sometimes enough to clear the alcohol away and resolve the problem, but medical attention might be necessary. Inhaling and swallowing it is generally dangerous and can be fatal. Exposure can be acute in which eye irritation, sore throat, headache, and dizziness might occur. Skin infections and other problems can result with chronic exposure.

A substance called methyl tertiary butyl ether can be metabolized into tertiary butyl alcohol. Safety measures should generally be in place for both when either one is used. The alcohol is usually stored in vented containers or closed drums, while inert gases can be vented above the containers to reduce the flammability of the air. Empty containers can also have explosive residue, and water is often ineffective at extinguishing fires that involve tertiary butyl alcohol. Carbon dioxide or dry chemicals are often used to put out such a fire.
AS FEATURED ON:
AS FEATURED ON:












Discuss this Article
Post your comments