At AllTheScience, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What Is Phosphorylation?
Phosphorylation is a chemical process in which a phosphate group (PO43-) is added to a compound. It normally applies to organic chemistry and is crucial to all living organisms. The process is involved in protein synthesis and in the production of adenosine
Normally, energy is required for the biochemical reactions that involve the addition of a phosphate group to a molecule. Often, this energy comes from ATP molecules. ATP contains three phosphate groups, one of which is easily removed. The removal of this group releases considerable energy, which can be used to enable a phosphorylation reaction in which the phosphate group is added to another molecule — for example, glucose. Thus, phosphate groups can easily be transferred from ATP to other molecules.

These reactions, however, require that ATP and the receptor molecule be brought together so that the transfer can take place. This is achieved by enzymes known as kinases. These are large, complex proteins, which may contain several hundred amino acids. The shape of the enzyme is crucial: the structure of a kinase enzyme is such that both the ATP and the receptor molecule can be accommodated in close proximity to allow the reaction to proceed. An example is glycerol kinase, which facilitates the transfer of a phosphate group from ATP to glycerol; this is part of the process that produces phospholipids, which are used in cell membranes.
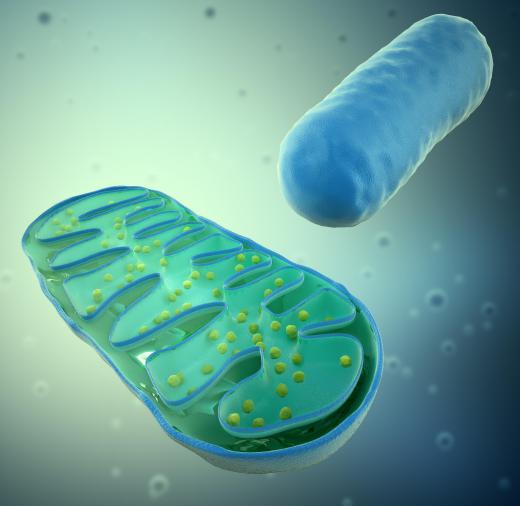
ATP is itself produced by a well-known phosphorylation process called oxidative phosphorylation, in which a phosphate group is added to adenosine diphosphate (ADP) to produce ATP. The energy for this process ultimately comes from the food we eat, but more specifically the oxidation of glucose. It is a very complex process with many steps, but in simple terms, the energy from glucose is used to form two compounds, known as NADH and FADH2, which provide the energy for the rest of the reaction. The compounds are reducing agents that easily part with electrons, so that they can be oxidized. Phosphate groups are added to ATP molecules using the energy released by the oxidation of NADH and FADH2; this reaction is facilitated by the enzyme ATP synthetase.

Many different kinases are found in both plants and animals. Due to their importance in so many cellular processes, a phosphorylation assay has become a common laboratory procedure. This involves testing samples of cell material to check if protein phosphorylation has taken place and, in some cases, measuring its extent. There are a number of different methods used to check for phosphorylation, including labeling phosphate groups with radioisotopes, the use of antibodies specific to the phosphorylated protein and mass spectrometry.
As of 2011, extra signal-regulated kinases (ERKs) — enzymes that are involved in signaling activities within the cell — are an area of particular interest. ERK phosphorylation plays a role in regulating various cell functions, including mitosis and other processes related to cell division. This process is relevant to some areas of cancer research as it can be activated by carcinogenic substances and by virus infections, leading to uncontrolled cell division and other cancer-related effects. Research into possible cancer treatments that involve inhibiting this process is underway. A phosphorylation assay can be used to test different substances for their effectiveness in this role.
AS FEATURED ON:
AS FEATURED ON:













Discuss this Article
Post your comments